One stop power supply solution
Power Supply Solution for In Vitro Diagnostic Devices
Safe, stable, and compliant power adapters tailored for IVD diagnostic equipment
We deliver optimized medical-grade power supply solutions specifically designed for in vitro diagnostic (IVD) devices such as biochemical analyzers, blood glucose meters, HbA1c analyzers, digital thermometers, and immunoassay systems. These power adapters meet international medical safety standards and offer the long-term stability and precision required for accurate diagnostics and reliable daily operation in clinical labs and point-of-care environments.

Power supply challenges and requirements for in vitro diagnostic systems
IVD equipment plays a crucial role in modern healthcare by enabling early disease detection and clinical decision-making. To maintain the integrity of test results, power supplies must deliver clean, stable output with minimal interference.
Common requirements include:
Ultra-low ripple and noise: to ensure accuracy in biochemical signal processing
Reliable voltage output: essential for continuous analyzer and sensor performance
IEC 60601-1 compliance: required for integration into medical-grade diagnostic systems
Robust EMC design: minimizes electromagnetic interference with nearby lab equipment
Worldwide voltage input and plug compatibility: supports international use in hospital and lab settings
Recommended Power Supplies for In Vitro Diagnostic Devices
Discover our high-performance medical power supplies tailored for:
Fully automatic biochemical analyzers
Blood glucose and HbA1c analyzers
Electronic blood pressure monitors
Fluorescence immunoassay systems
Digital thermometers and flat panel detectors
Medical sprayers and enema machines
These power solutions offer:
✅ Low ripple and noise for high-precision testing and diagnostics
✅ Consistent voltage regulation for 24/7 lab operation
✅ High safety and reliability, even in complex analytical environments
✅ Flexible customization—DC connectors, cables, and housing designs tailored to your devices
Core advantages of our power products
✅ Certified to IEC 60601-1: compliant with global medical safety standards
✅ Wide voltage output range (12V–48V): to support a broad spectrum of IVD systems
✅ Excellent EMC performance: meets EN55011 Class B, ensuring minimal interference
✅ Compact and durable: supports integration in space-constrained diagnostic devices
✅ Full OEM/ODM customization available: adapt plug types, output specs, and housing to your application
Application scenarios and customer examples
Our IVD power solutions are widely deployed in:
🔬 Clinical laboratories and pathology centers
🏥 Hospital diagnostic departments
🧪 Point-of-care testing (POCT) and portable analysis kits
They are trusted by global diagnostic brands and healthcare OEMs for high performance and safety compliance.
Certification and technical assurance
All Quankang IVD power adapters are rigorously tested and certified to meet international standards:
Medical compliance: IEC 60601-1
Safety certifications: CE / CB / UL / FCC / PSE / UKCA
EMC compliance: EN55011 Class B
Environmental standards: RoHS / REACH
We also provide complete technical documentation:
Product datasheets
Medical and safety certificates
System selection and integration support
Featured Products

Medical External Power Supply UES06WNCP1-SPA
Home Wall Plug Medical External Power Supply UES06WNCP1-SPA Series 12V AC to DC Wall Plug Medical External Power Supply USB Type Switching Power Adapter 6

Medical External Power Supply UES06WZ-SPA
Home Wall Plug Medical External Power Adapter UES06WZ-SPA 6W 12V Medical Wall Plug External Power Supply Medical Adapter Interchangeable AC Plugs 6 W 90 ~
Featured Resources
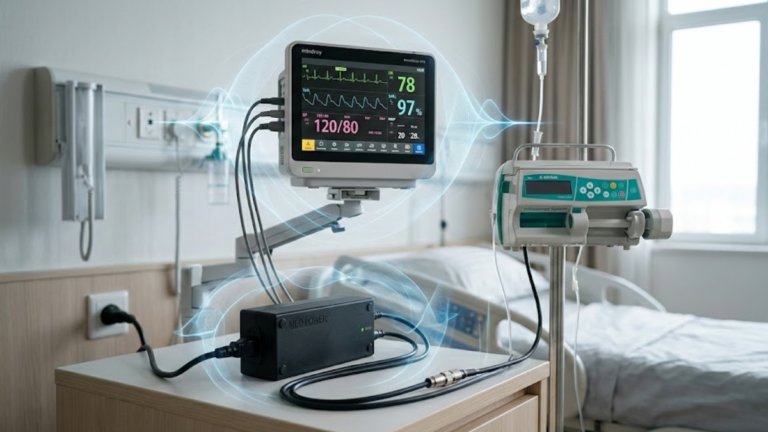
Introduction: EMC Is a Safety Requirement, Not a Performance Feature In medical electrical equipment, electromagnetic compatibility (EMC) is not merely about avoiding interference—it is a

1xMOPP vs 2xMOPP in Medical Power Supplies Isolation requirements in medical power supplies are a cornerstone of IEC 60601-1 compliance. Understanding the distinction between 1xMOPP (Means

Introduction: Certification Failures Rarely Start at the Test Lab Most medical device certification failures are not caused by last-minute mistakes.They originate months earlier—often at the
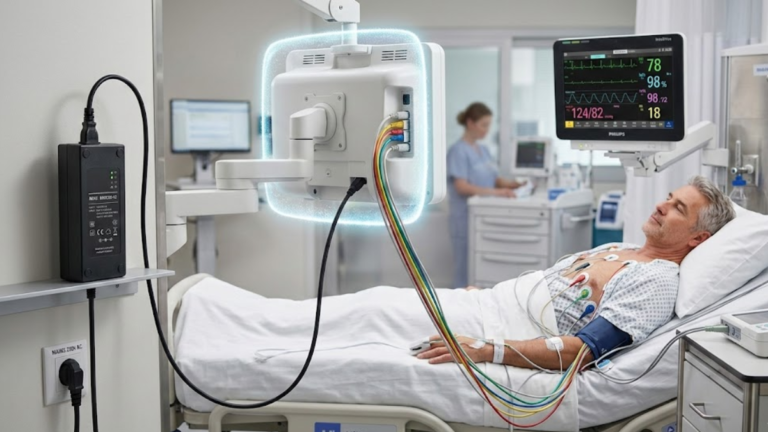
Introduction: Patient Connection Changes Everything In medical device design, patient connection is a regulatory boundary, not a functional feature.Once a device is classified as patient-connected,

Introduction: Power Supply Selection Is a Certification Decision medical power supply selection is often treated as a late-stage electrical task. In reality, it is one of
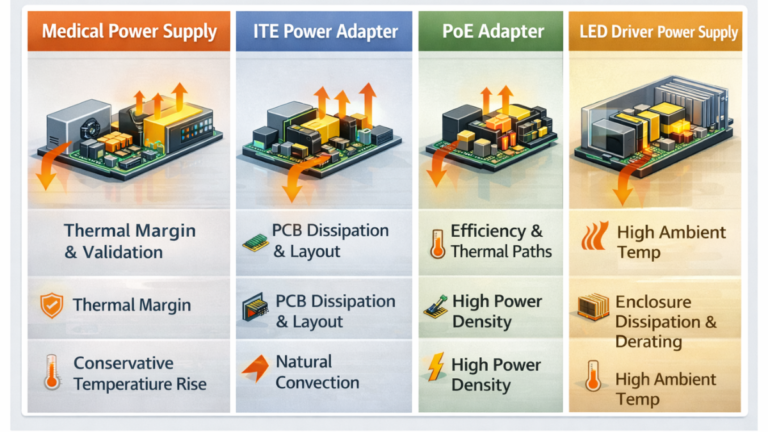
In the process of AC-DC power supply, smaller and higher power density development, the thermal design becomes a cord problem affecting the product’s reliability and


