Choosing the right power adapter for medical equipment is rarely as simple as matching a voltage number. Engineers often have to consider electrical performance, safety certification, connector compatibility, and long-term reliability before a device can enter mass production.
A poorly selected adapter may lead to unstable startup behavior, overheating, certification failure, or even product redesign. Because of this, understanding how to select a reliable medical power adapter is an essential step in medical device development.
This guide explains the key parameters engineers should evaluate when selecting a medical power supply solution for healthcare equipment.
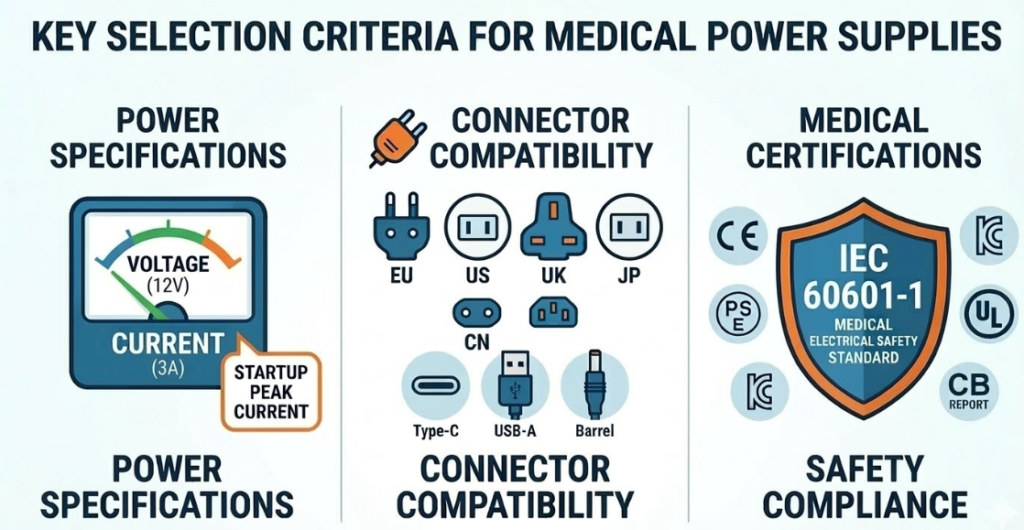
Three Key Factors When Selecting a Medical Power Adapter
When engineers evaluate a power adapter for medical devices, three parameters usually define the first screening stage:
- Power specifications (voltage and current)
- Connector compatibility (AC plug and DC plug)
- Medical safety certifications
Once these three parameters are confirmed, engineers can begin comparing different AC-DC medical power adapters that match the electrical and regulatory requirements of the device.
Power Requirements: Voltage and Current
The electrical rating of a power adapter is defined by its output voltage and current.
For example, a 36 W medical power adapter may provide:
12 V / 3 A
9 V / 4 A
Before selecting an adapter, engineers must first determine the required output voltage and operating current of their device.
However, the nominal current requirement is not always the full story. Many electronic devices require a higher current during startup, a phenomenon known as startup peak current.
This startup peak may last only a fraction of a second, but it can still cause serious issues if the power supply cannot handle it. A weak adapter may experience voltage drop, protection triggering, or repeated restart loops during device startup.
A practical rule is simple: match the voltage exactly and allow additional headroom for current.
If your device requires 12 V / 3 A, selecting a 12 V / 3 A adapter that barely meets the specification may lead to unstable startup behavior. Choosing an adapter with extra current margin improves system stability.
Connector Compatibility: AC Plug and DC Plug
Power connectors are another critical factor in adapter selection.
The AC plug determines where the adapter can be used geographically. For example:
- EU plug for Europe
- US plug for North America
- UK plug for the United Kingdom
Different regions also have different socket depths and mechanical tolerances. Using the wrong plug design may cause poor socket contact or unstable power connection during vibration tests.
DC connectors are even more sensitive. A connector that appears visually correct may still fail if the inner pin diameter differs by only 0.1 mm, if the polarity is reversed, or if the contact spring provides insufficient pressure.
In many cases, devices fail not because the internal electronics malfunction, but because poor connector contact resistance leads to intermittent power supply.
For this reason, engineers should confirm the following parameters when selecting DC connectors:
- Connector size
- Polarity
- Insertion depth
- Cable angle
- Cable flexibility
- Plug-unplug cycle frequency
Modern medical power adapter solutions can support interchangeable AC plugs and customized DC connectors such as Type-C, USB-A, or standard barrel connectors.
Medical Certifications and Safety Compliance
Unlike consumer electronics power supplies, medical adapters must comply with strict safety standards.
The most important certification is IEC 60601-1 medical electrical safety standard, which defines electrical safety requirements for medical equipment and related power supplies.
Many manufacturers obtain a CB report under this standard, which allows them to apply for regional certifications more easily.
For example:
- Europe requires CE certification based on EN 60601-1
- Japan requires PSE certification
- Korea requires KC certification
- North America commonly requires UL approval
Confirming these certifications early helps prevent delays during medical device regulatory approval.
Final Evaluation: Reliability and Stress Testing
Matching electrical specifications and certifications is only the first step in selecting a power supply.
Real-world testing is essential to ensure that the adapter performs reliably under operating conditions.
Engineers typically evaluate the following factors:
- Startup behavior
- Load fluctuation performance
- Temperature rise during operation
- Long-term stability under continuous load
Additional tests such as aging tests, drop tests, lifecycle analysis, and thermal margin evaluation help verify whether the adapter will remain reliable after large-scale production begins.
Choosing a stable medical power supply early in the design stage can prevent redesign cycles and certification delays later in product development.

Conclusion
Selecting the right power adapter for medical devices requires more than simply matching voltage and current specifications.
Engineers must also evaluate connector compatibility, regulatory certification, and long-term reliability under real operating conditions.
A well-designed medical power adapter helps ensure stable device operation, smoother certification processes, and reduced risk during mass production.
If you are designing medical equipment and evaluating power solutions, exploring reliable medical power adapter solutions can help improve both product stability and development efficiency.
Contact us to get a better-designed power supply solution.